Clinical Trials
01.
About
Clinical trials are crucial to demonstrating the efficacy of our products for the treatment of chronic pain and other neurological conditions. Details of our current clinical trials are outlined below and follow our successful proof of concept trial conducted in the UK in 2020/2021.
In the UK study, conducted from mid 2020 until early 2021, patients reported significant reductions in their levels of pain as well as in anxiety and depression. At the same time, they reported improvements in sleep, mood and quality of life. The full trial results were published in medical journal, Frontiers of Pain.
We are not recruiting for clinical trials at the moment
02.
Current Clinical Trials
Waitematā District Health Board and Auckland University of Technology chronic pain trial
We are thrilled to have recently achieved the target of recruiting 116 patients in the New Zealand trial partnership with Auckland University of Technology (AUT) and Waitematā District Health Board.
The NZ trial is a random Controlled Clinical Trial which is considered the gold standard of clinical trials. All initial patient treatments are scheduled to be completed in early October and initial results will be available shortly after.
There will be 12 months post-trial follow up of the trial participants to provide the company with a rich dataset and understanding of EEG neurofeedback and a patient’s journey on the Axon system.
UK NHS Trial – Chronic Pain
This trial has received ethical approval from the NHS and we are scheduled to start recruiting patients on 1 September 2022. This trial will run for 20 weeks and is designed to harness information about the ‘chronic pain brain’ before and after EEG neurofeedback treatment as well as serving as a pre-purchasing trial for the NHS East Kent hospital.
To register for future clinical trials, please click here.
01.
Trial Results

Selected Trial Results
As well as thousands of hours of testing Axon has undergone three clinical trials with a total of 150 patients consisting of 12,000 treatments delivered and billions of lines of data analysed. The results of the trials are pretty conclusive, Axon is safe and effective with 94% of patients responding to treatment, 80% had a reduction in Pain and 65% had improvements in Mental Health, Anxiety and Depression as well as Sleep.
Below are some of the selected results from the trials:
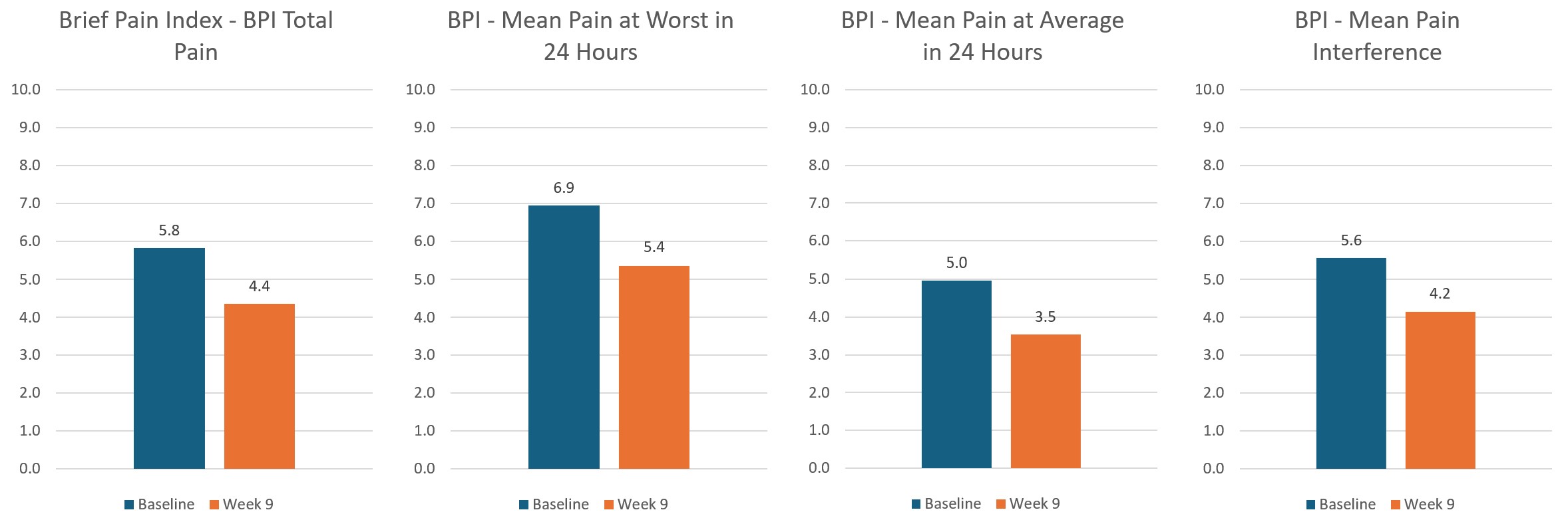
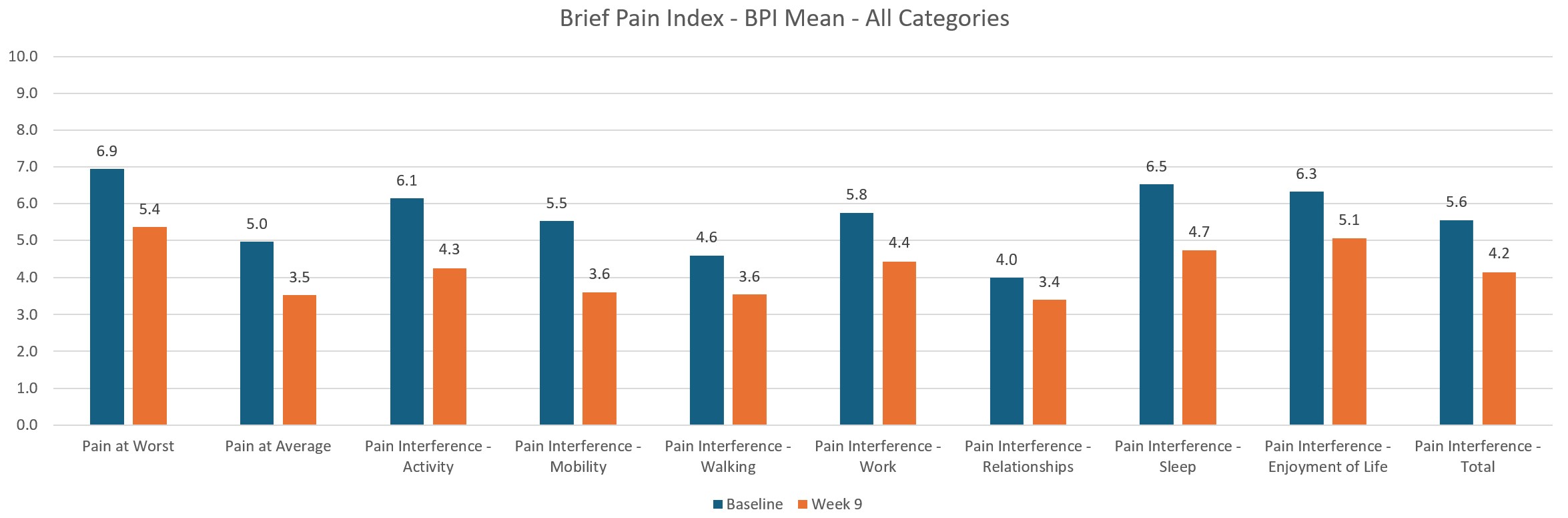
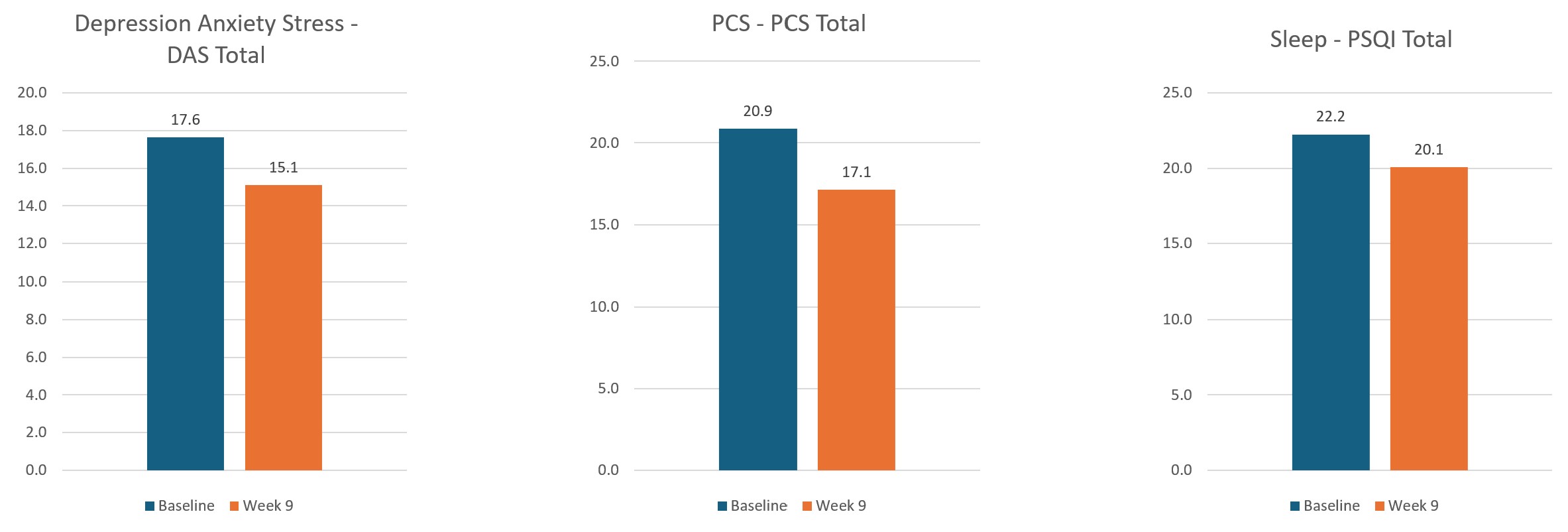
Upcoming Auckland
Chronic Pain Trial
Auckland Trials
If you are interested in taking part in the Chronic Pain trial with the Waitemata District Health Board and live in the Upper North Island of New Zealand, then please contact the team.
For sales enquiries and trials outside Auckland
For all other trials and sales enquiries please contact the team at info@exsurgo.com.
Media
Chronic Pain Study (UK)
A recent UK study has confirmed the exciting potential of Exsurgo’s breakthrough treatment for chronic pain management.
In the UK study, conducted from mid-2020 until early 2021, patients reported significant reductions in their levels of pain, anxiety and depression. At the same time, they reported improvements in sleep, mood and quality of life. Furthermore, the improvements from the eight weeks of neurofeedback training were sustained at follow-up points – 4, 12 and 26 weeks. The full results of the trial are expected to be published shortly in a medical journal.
The UK study was intentionally small-scale, involving 16 patients, to serve as a proof-of-concept for the Axon system and lay the groundwork for further research. A much larger clinical trial is due to start in New Zealand shortly.
Trial Publications:
16 Patient Home Based Axon Intervention for Chronic Pain
UK PoC Clinical Trial:
https://www.frontiersin.org/articles/10.3389/fpain.2022.855493/full
116 Patient Home Based Axon intervention for Chronic Pain
Clinicial Trial in New Zealand
In review – to be published soon
10 Patient Home Based Axon intervention for Chronic Pain
UK NHS Clinical Trial
In review – to be published soon